Patient's benefits
- Protective barrier effect against allergens
- Reduction of nasal symptoms such as nasal congestion, sneezing, nasal irritation and rhinorrhea within 7 days
- Reduced eye symptoms such as redness, tearing and burning within 7 days
- Improvement in quality of life from the reduction of both nasal and ocular symptoms
- Reduced need for allergic rhinitis medication medication
- Improved patient well-being and sleep quality
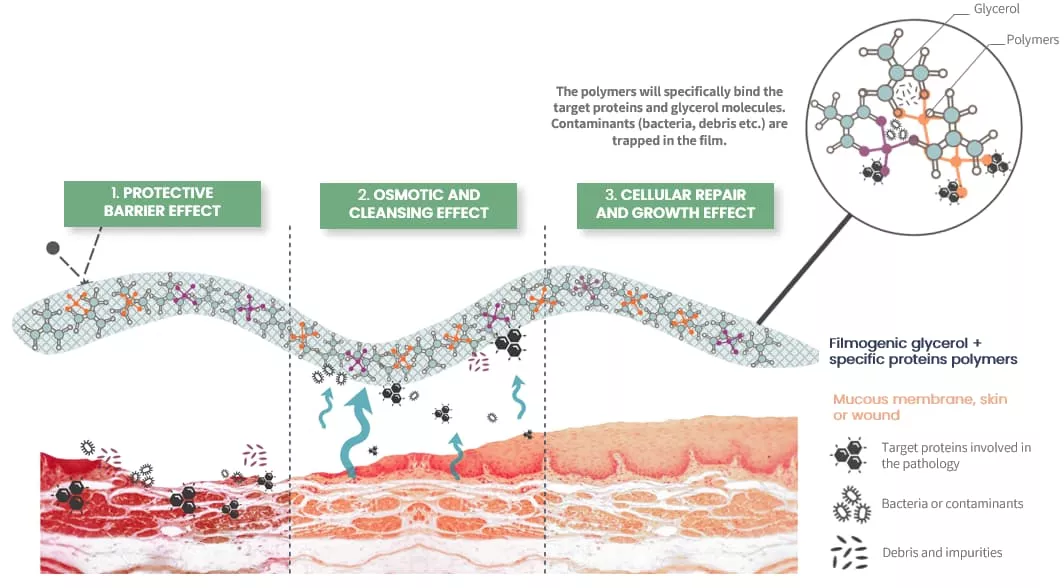
Mode of Action
- Protective Barrier: Establishes a filmogenic shield on the nasal mucosa, preventing irritants from triggering allergic reactions.
- Mechanical Cleaning: Utilizes osmotic activity to remove contaminants, reducing inflammation and symptoms.
- Cellular Repair: Promotes healing and regeneration of the nasal mucosa for sustained relief.
Clinical study
A randomized, double-blind clinical trial (n = 51) confirmed the strong efficacy of the product in relieving allergic rhinitis symptoms.
Within just 15 days, the treatment achieved a 96% reduction in total nasal symptoms and a 99% reduction in ocular symptoms, with noticeable effects as early as day 2. Additional improvements included a 73% decrease in rhinorrhea and 67% reduction in sneezing within the first week.
These results highlight the product’s rapid action, broad symptom relief, and significant improvement in quality of life for patients with confirmed allergic profiles.
Product Details
- Indication: Used in children for the symptomatic treatment of seasonal and intermitent mild-to-moderate allergic rhinitis.
- Composition: Made from plant-based ingredients
- Presentation: Nasal spray 15ml.
- Class: MDR Class IIa.
- Age: 3+.
- Storage conditions: Below 25°C.
- Legal manufacturer: VITROBIO (France).
- Product code: AEE.
Need more information
 This image is a representation. Your design will be featured in the final product.
This image is a representation. Your design will be featured in the final product.